ZnO is a wide band-gap semiconductor with unique physicochemical properties. This material is high-energy radiation and ambient atmosphere resistant and is relatively non-toxic to living organisms. ZnO alloyed with transition metals promises an above-room-temperature ferromagnetism and hence wide applicability in semiconductor spintronics. Its high exciton binding energy of 60 meV allows to reduce the lasing threshold and increases the efficiency of ultraviolet luminescence at room temperature. Despite these great advantages, development of ZnO-based electronics is strongly limited by the lack of large crystals of high quality suitable for industry as substrates for the homoepitaxy.
The main goal of this work is a comprehensive investigation of physical properties of ZnO bulk crystals obtained as by-product of zinc white production in Oława Foundry [1]. A very intriguing aspect of these crystals is their sheer size, as far as we know, exceeding the typical size of commercially available ZnO. The crystals which are the subject of this paper are about 1 cm wide and about 12 cm long rods of a hexagonal basis. They are transparent and reddish.
In order to verify the purity of ZnO crystals a number of experimental methods were applied. The energy-dispersive X-ray (EDX) spectroscopy analysis have shown that the crystal consist of Zn and O only (the concentration of possible impurities is below the detection limit of EDX method). The powder X-ray diffraction measurements have demonstrated a hexagonal wurtzite structure of the samples and yielded no additional peaks resulting from possible precipitates or other crystal phases. The crystal orientation was determined by the Laue method. The crystal face parallel to its axis was analyzed at several points along the length of the sample. The investigated face corresponds to a (110) orientation, thus the crystals’ axis coincides with the wurtzite c-axis, and the investigated crystal has the same orientation throughout its length. In order to check are the structure properties of investigated single crystals uniform or not the intensity of the Bragg peak 200 was measured in several places along the sample surface on the distance exceeding 10 mm. The analysis of the intensity distribution maps in the reciprocal space demonstrated a very homogeneous distribution of defects and small scattering of the mosaic block orientation in a big sample area. The analysis of the 110 Bragg peak intensity distribution, obtained by the neutron diffraction with the use of modern 2D neutron detector have demonstrated that the sample has indeed a form of a single crystal – see Figure 1(a). In order to determine the linear expansion coefficients for the a and c parameters the X-ray powder diffraction was performed with the use of synchrotron radiation in the temperature range from 15 K to 1100 K at Hasylab (the diffraction pattern obtained at 20 K is exemplified in Figure 1 (b)).
Samples were characterized by various optical methods: photoluminescence (PL), cathodoluminescence (CL), Raman scattering (RS), and optical absorption (OA). The as grown crystals have strong red absorption that can be related to Fe or Mn impurities. The PL spectra have exhibited broad defect-related green luminescence (GL) band – see Figure 2. The GL band is very wide, indicating a presence of a variety of defects in these crystals. The room temperature time-resolved PL have revealed that the life-time of near band-gap luminescence (NBL) and GL is 60 – 110 ps and 6 – 12 ns, respectively. Short lifetime of NBE indicates fast recombination on defects. The single-phonon excitations were studied by the infrared reflectivity and by the first-order RS. Infrared plasma reflection measurements yielded the magnitude of the electron concentration of 2.7 x 1016 cm-3 what was roughly confirmed by the Hall effect measurements.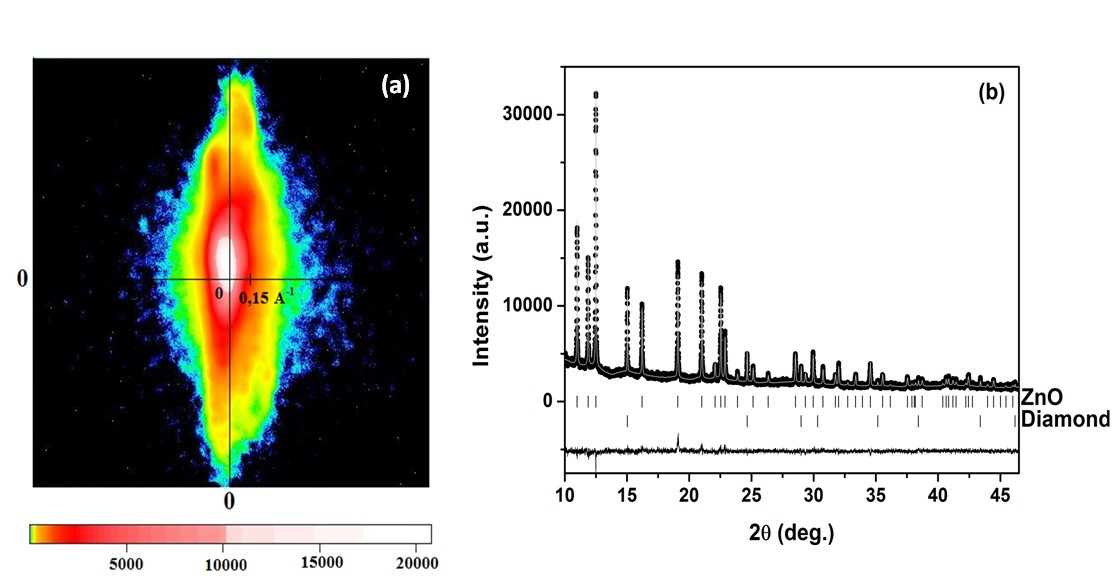 Figure 1. (a) The intensity distribution for 110 Bragg peak for the whole crystal obtained by the neutron diffraction with the use of a 2D detector. (b) Rietveld refinement plots for ZnO sample at 20 K. The observed data are indicated by dots and the calculated ones by the solid gray line. The vertical bars mark the positions of Bragg reflections, whereas the lower curve represents the difference between the observed and calculated powder diffraction patterns. The used X-ray wavelength was about 0.53836 Å. Figure 1. (a) The intensity distribution for 110 Bragg peak for the whole crystal obtained by the neutron diffraction with the use of a 2D detector. (b) Rietveld refinement plots for ZnO sample at 20 K. The observed data are indicated by dots and the calculated ones by the solid gray line. The vertical bars mark the positions of Bragg reflections, whereas the lower curve represents the difference between the observed and calculated powder diffraction patterns. The used X-ray wavelength was about 0.53836 Å.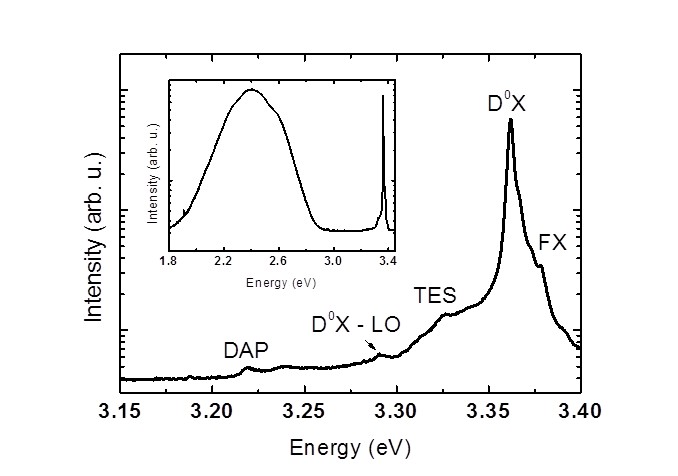 Figure 2. The photoluminescence spectrum of ZnO crystal at T = 5 K. Figure 2. The photoluminescence spectrum of ZnO crystal at T = 5 K.
The studies were partially supported, by the European Union within the European the European Regional Development Fund, through grant Innovative Economy (POIG.01.01.02-00-008/08), and by the Polish National Science Centre grant No: DEC-2011/03/B/ST3/02664. Portions of this research were carried out at the light source DORIS III at DESY, a member of the Helmholtz Association (HGF). This work was partially supported by EC under contract II-20100155. [1] J.W. Nowak R.S.W. Braithwaite, J. Nowak, K. Ostojski, M. Krystek, W. Buchowiecki, Journal of Gemmology 30, 257 (2007). |