| Search for content and authors |
Antiproliferative activity of novel acetylenic derivatives of betulin against G-361 human melanoma cells |
| Ewa B. Bębenek 1, Ewa Chodurek 2, Arkadiusz Orchel 2, Zofia Dzierżewicz 2, Stanisław Boryczka 1 |
|
1. Department of Organic Chemistry, The Medicial University of Silesia, Jagiellońska 4, Sosnowiec 41-200, Poland |
| Abstract |
Malignant melanoma is highly resistant to chemotherapy and radiotherapy, particularly in the metastatic or relapse phase. Median survival time of metastatic patients is 6-9 months and the 5-year survival rate does not exceed 1-2%. Therefore, new possible treatment strategies are still searched for. Inhibition, delay or reversal of carcinogenesis using natural or semisynthetic compounds seems to be a promising approach [1]. Betulin (1) is a natural compound with cytotoxic activity against cancer cells. It can be easily converted to various semisynthetic derivatives [2-3]. The aim of the study was to investigate the effect of acetylenic derivatives of betulin (2-7) on cell proliferation in G-361 human melanoma cell line. To assess cell proliferation, G-361 cells were fixed with 10% trichloroacetic acid and stained with sulforhodamine B. The range of concentrations of the tested compounds was from 1 to 20 µg/mL and the cells were treated for 72h. Monoesters 2-4, obtained by a replacement of the hydroxyl group at C-28 position of betulin (1) by alkynyl groups, exhibited the most potent cytotoxicity. Instead, simultaneous esterification of the C-3 hydroxyl group (diesters 5-7) completely abolished the cytotoxic action of the compound (in a concentration range: 1-20 μg/mL). 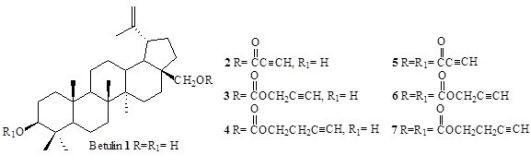
References: [1] Weller R, Hunter J, Savin J, Dahl M. Dermatologia kliniczna. Lublin: Czelej; 2011: 318. [2] Tolstikov G A, Flekhter O B, Shultz E E, Baltina L A, Tolstikov A G. Betulin and its derivatives. Chemistry and biological activity. Chem Sustain Develop; 2005: 13, 1–29. [3] Boryczka S, Bębenek E, Wietrzyk J, Kempińska K, Jastrzębska M, Kusz J, Nowak M. Synthesis, structure and cytotoxic activity of new acetylenic derivatives of betulin. Molecules; 2013: 18, 4526-4543. |
| Legal notice |
|
| Related papers |
Presentation: Poster at IX Multidyscyplinarna Konferencja Nauki o Leku, by Ewa ChodurekSee On-line Journal of IX Multidyscyplinarna Konferencja Nauki o Leku Submitted: 2014-03-13 12:50 Revised: 2014-05-02 09:57 |