| Search for content and authors |
Capecitabine molecular structure in the liquid state as predicted from NMR measurements and theoretical calculations. |
| Piotr Cmoch 1,2, Piotr Krzeczyński 1, Andrzej Leś 1,3 |
|
1. Instytut Farmaceutyczny (PRI), Rydygiera 8, Warszawa 01-793, Poland |
| Abstract |
In spite of enormous literature (over 6800 papers) reporting biochemical, pharmacological or medicinal data, the molecular structure of capecitabine was rarely described. Scarce spectral and crystal data are not consistent with respect to the tautomeric forms occuring in solid or liquid phases. This has prompted us to study capecitabine in DMSO-d6 (at room temperature) and THF-d8 (at various temperatures) solutions. Based on 1H, 13C, 15N and 19F NMR (HSQC, HMBC) spectra it was found that at least two tautomeric forms (I-amino, II-imino) exist in equilibrium. 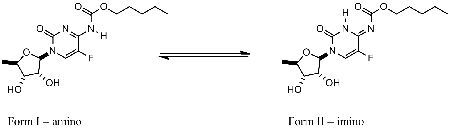
At T=218 K form I-amino dominates. It was also found (1H, 19F spectra) that tautomeric equilibrium is temperature dependent in various solvents. Density functional theory quantum mechanical calculations suggested that tautomeric form I-amino is prefered for series of molecules related structurally to capecitabine and posessing the central moiety reminding 5-fluorocytosine. However, if the intramolecular and intermolecular hydrogen bond can be formed (due to conformational changes or interactions with neighbouring molecules) tautomeric form II-imino can be prefered. The computational part was performed at the ICM Warsaw University computer center within the G18-6 grant. |
| Legal notice |
|
| Related papers |
Presentation: Poster at IX Multidyscyplinarna Konferencja Nauki o Leku, by Piotr KrzeczyńskiSee On-line Journal of IX Multidyscyplinarna Konferencja Nauki o Leku Submitted: 2014-03-13 14:55 Revised: 2014-04-29 12:50 |