| Search for content and authors |
Theorems for Numerical Simulation of Solution Growth with Segregation and Fluid Flow |
| Hiromoto Susawa |
|
Home, Aichi 489-0888, Japan |
| Abstract |
1. Introduction
7. Assumptions related to fluid flow The model is in the xz plane. DL > DM. The fluid flow traverses the boundary layers of mole fractions. Velocity perpendicular to the boundary of fluid flow is zero. Discussion starts with the mole fractions without the fluid flow. 8. Theorems by generalizing a series of these studies [5-9] 8.1 Enhancement of growth by fluid flow Case 1: A liquid solution retaining the initial supersaturated state in an outer region of the boundary layers of XL and XM flows to their boundary layers. ———— Theorem 2: Theorem 2 deals with Case 1 and assumes the base of model and assumptions related to fluid flow. Then, the fluid flow increases XL( x, z, t ) more than XM( x, z, t ) around the border between their boundary layers and the outer region of the boundary layers. Reason: Fig. 1b is an example to show the profiles of mole fractions in their boundary layers. Fig. 2 characterizes the boundary layers. This figure defines positions, e and f. Position e is in an outer region of the boundary layers. Position f is inside the boundary layer of XL and outside the boundary layer of XM. Suppose that a block of liquid solution retains the initial supersaturated state at Position e and t = t1, and comes to Position f at t = t2. Fig. 2a illustrates how the fluid flow affects XL. The fluid flow transports a block of liquid solution in which XL has XLb to the boundary layer. A1 = A2 + A3. XL( x, Position f, t2 ) increases by the amount proportional to Area A2. Fig. 2b illustrates the effect on XM. The relation between Area A4, A5 and A6 is A4 = A5 + A6. The fluid flow increases XM( x, Position f, t2 ) by the amount proportional to Area A5. The effect on XM is smaller than the effect on XL because the profile of XM was more saturated to its initial value than that of XL at t1. Obviously, the above geometric discussion can be expanded to three-dimensional space. The upstream position and destination do not need to be along the z-axis. ———— The following becomes an index to verify the computed results. It is obvious form Fig. 2. Theorem 3: Theorem 3 uses the assumptions and result of Theorem 2. The fluid flow decreases δL and δM. ———— From Theorem 3, this fluid flow enhances the growth. The following is a preparation for the next theorem. Proposition 1: The fluid flow increases XL and decreases XM on the growth interface. ———— Theorem 4: Theorem 4 uses the assumptions and results of Theorem 2 and 3. Theorem 4 assumes the result of Theorem 2 propagates to the growth interface and dXM / dXL < 0 on the liquidus line. Proposition 1 is valid under Eq. 1. Reason: From Theorem 3, pL and pM increase. The increase of XM is less than that of XL in the vicinity of growth interface because the result of Theorem 2 propagates to the growth interface. The left hand side of Eq. 1 is greater than the right hand side if the equilibrium state remains constant. On the other hand, on the growth interface, XL and XM are on the liquidus line; then, when XL increases, XM decreases. The increase of XL on the growth interface contributes to the decrease of pL. The decrease of XM on the growth interface contributes to the increase of pM. That is, this change of equilibrium state contributes to the adjustment of the left hand side of Eq. 1 to the right hand side. Fig. 4a is examples of NL profiles in the boundary layers of XL. These profiles show how XL varies in z direction. Fig. 4b shows NL in the vicinity of growth interface. On the growth interface, “dYL / dXL > 0 or dYM / dXM > 0” is obvious. Suppose XL increases on the growth interface. Then, YL increases and YM decreases. YL >> XL. YM >> XM. Then, the right hand side in Eq. 1 increases. Therefore, the increase of XL on the growth interface decreases (the left hand side) - (the right hand side). At a certain value of XL on the growth interface, the left hand side balances with the right hand side. When suppose XL decreases on the growth interface, by repeating the similar consideration, we find Eq. 1 is not satisfied. That is, Proposition 1 is valid under Eq. 1. 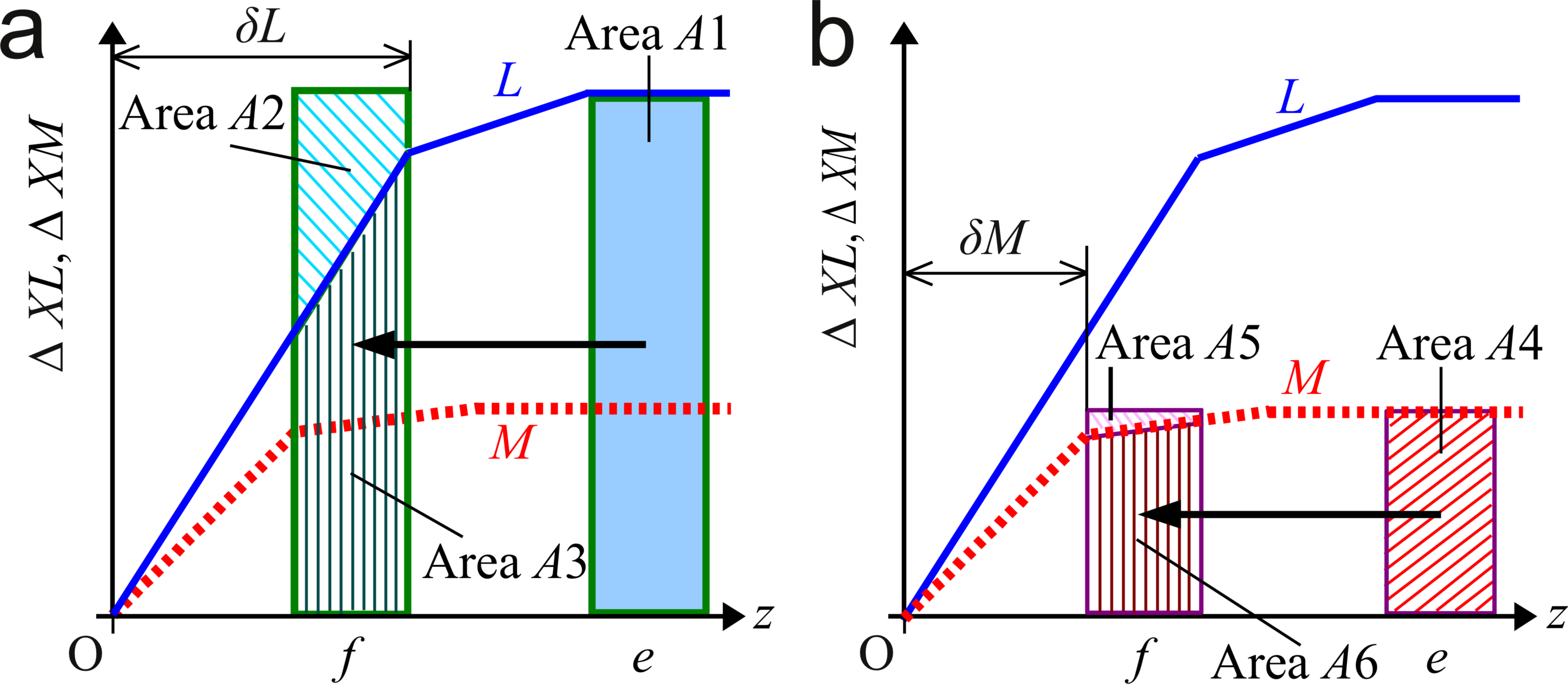 Fig. 2. Schematic illustration of the effect on the boundary layers by a fluid flow. Position f is inside the boundary layer of XL and outside that of XM. Suppose a liquid solution retaining the initial supersaturated state flows to Position f : (a) Effect on the boundary layer of XL, (b) Effect on the boundary layer of XM. 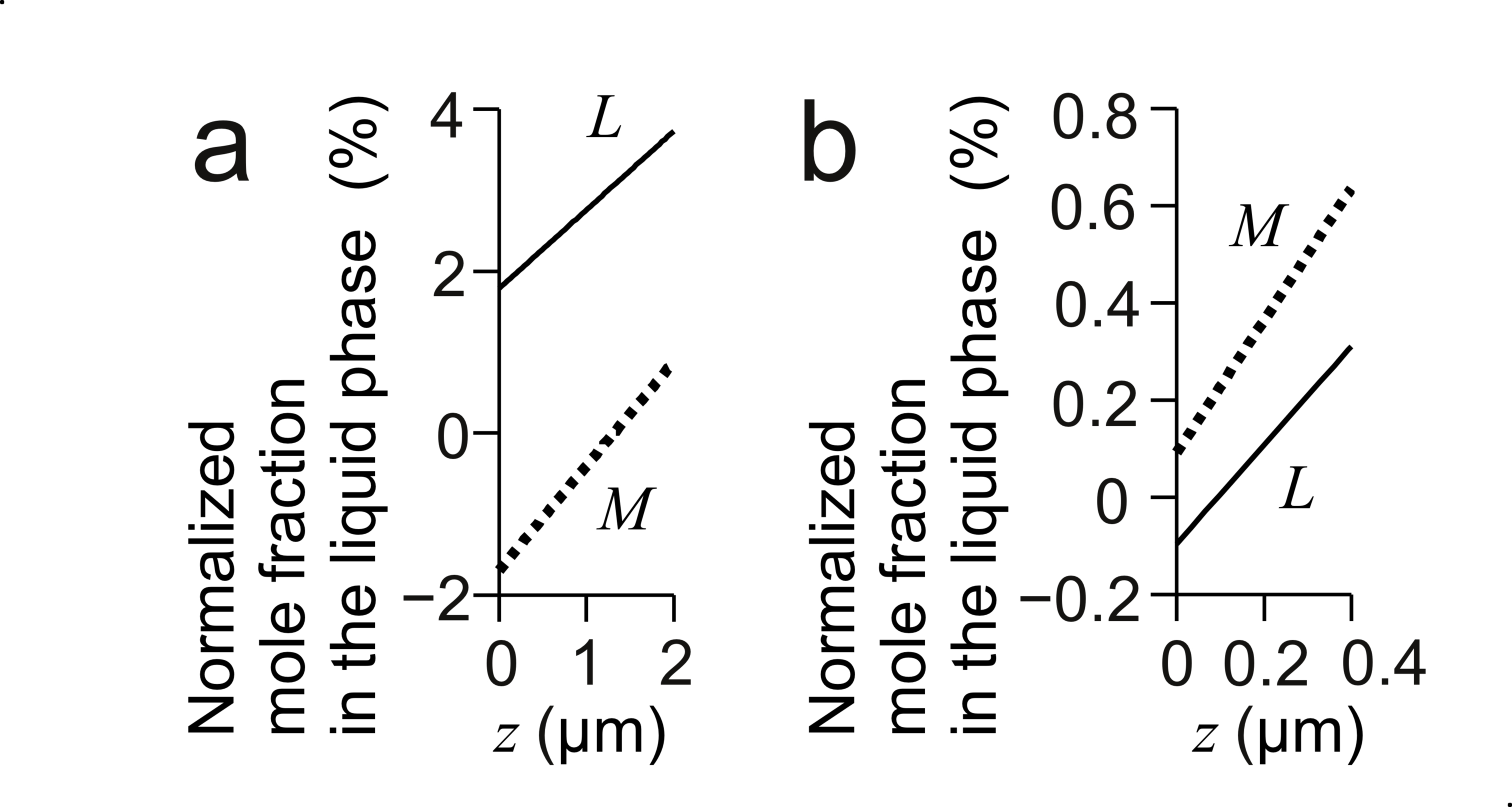 Fig. 3. Examples of normalized mole fractions in the liquid phase on the line, x = 0 near the growth interface Concrete model is the same as that in Fig. 1b. (a) The enhancement of growth by fluid flow at t = 0.278 s. (b) The inhibition of growth by fluid flow at t = 0.12 s. 8.2 Inhibition of growth by fluid flow Case 2: The direction of a fluid flow is from the growth interface to an outer region of the boundary layers constituted by the mole fractions of solutes. ———— Case 2 is opposite to Case 1. We find out the reasons with the similar procedures. The fluid flow inhibits the growth. We move the control volumes form Position f to Position e in Fig. 2. The conclusions are opposite to those in Case 1. Theorem 5: Theorem 5 deals with Case 2 and assumes the base of model and assumptions related to fluid flow. Then, the fluid flow decreases XL( x, z, t ) more than XM( x, z, t ) around the border between their boundary layers and the outer region of the boundary layers. Theorem 6: Theorem 6 uses the assumptions and result of Theorem 5. The fluid flow increases δL and δM. Proposition 2: The fluid flow decreases XL and increases XM on the growth interface. Theorem 7: Theorem 7 uses the assumptions and results of Theorem 5 and 6. Theorem 7 assumes the result of Theorem 5 propagates to the growth interface and dXM / dXL < 0 on the liquidus line. Proposition 2 is valid under Eq. 1. References [1] H. Susawa, M. Hirotani, T. Kato, Surface Emitting LED with Reflector on GaAs, 1989 fall 50th meeting held by Jpn. Soc. Appl. Phys. 28 p-ZB-10 (in Japanese) [2] T. Kato, H. Susawa, M. Hirotani, T. Saka, Y. Ohashi, E. Shichi and S. Shibata, J. Cryst. Growth, 107 (1991) 832. [3] N. Yamauchi, T. Saka, M. Hirotani, T. Kato, and H. Susawa, European Patent Office, Patent Number: EP0483868B1 (Issue Date: January 22, 1997). [4] H. Susawa, T. Tsuji, T. Jimbo, T. Soga, J. Chem. Eng. Jpn. 40 (2007) 928. [5] H. Susawa, T. Tsuji, K. Hiramatsu, T. Jimbo, T. Soga, Simulation of InGaP Liquid Phase Epitaxy Including Convection, Theor. Appl. Mech. Jpn. 55 (2006) 279-284. [6] H. Susawa, T. Tsuji, K. Hiramatsu, T. Jimbo, T. Soga, DOI http://dx.doi.org/10.2197/ipsjdc.3.114. [7] H. Susawa, Numerical simulation of compositional variation in liquid phase epitaxy focusing on the forced convection in the melt, 16th Int. Conf. Cryst. Growth (2010) Sess.: 01 Aug. 11. [8] H. Susawa, Leibniz Inst. Cryst. Growth (IKZ), abstract book 5th Int. Workshop Cryst. Growth Technol., Berlin, (2011) 61. [9] H. Susawa, Proc. 7th Int. Workshop Model. Cyst. Growth, Dep. Chem. Eng., Natl. Taiwan Univ., (2011) 103. [10] Panel discussion at the 55th Natl. Congr. Theor. Appl. Mech. in Kyoto Jpn. (2006). [11] K. Hiramatsu, S. Tanaka, N. Sawaki, I. Akasaki, Jpn. J. Appl. Phys. 24 (1985) 1030. [12] I. Crossley, M.B. Small, J. Cryst. Growth 15 (1972) 268. [13] M. Feng, L.W. Cook, M.M. Tashima, G.E. Stillman, J. Electron. Mater. 9 (1980) 241. |
| Legal notice |
|
| Related papers |
Presentation: Poster at 17th International Conference on Crystal Growth and Epitaxy - ICCGE-17, General Session 1, by Hiromoto SusawaSee On-line Journal of 17th International Conference on Crystal Growth and Epitaxy - ICCGE-17 Submitted: 2013-04-12 16:55 Revised: 2013-06-23 20:44 |